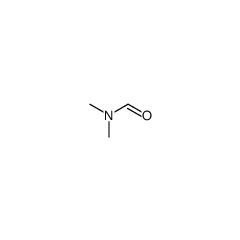
Two methyl formamide (English: dimethylformamide, abbreviated DMF) is a transparent liquid that can be miscible with water and most organic solvents. It is a common solvent for chemical reactions. Pure two methyl formamide is odorless, but industrial grade or metamorphic two methyl formamide has fishy smell, because it contains two methyl amine impurity. The name is derived from the fact that it is a two-methyl substitute for formamide (formic acid amide) and two methyl are located on N (n) atoms. Two methyl formamide is a high boiling point of the polar (hydrophilic) non-proton solvent, can promote the SN2 reaction mechanism. Two methyl formamide is made using formic acid and two methyl amine. Two methyl formamide is unstable (especially at elevated temperatures) in the presence of strong alkali such as sodium hydroxide or strong acid such as hydrochloric acid or sulfuric acid, and is hydrolyzed to formic acid and two methyl amine.