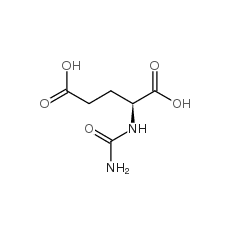
Carglumic acid is an orphan drug, marketed by Orphan Europe under the trade name Carbaglu. Carglumic acid is used for the treatment of hyperammonaemia in patients with N-acetylglutamate synthase deficiency. The initial daily dose ranges from 100 to 250mg/kg, adjusted thereafter to maintain normal plasma levels of ammonia. The US FDA approved it for treatment of hyperammonaemia on March 18, 2010. Orphan Drug exclusivity expires on March 18, 2017.